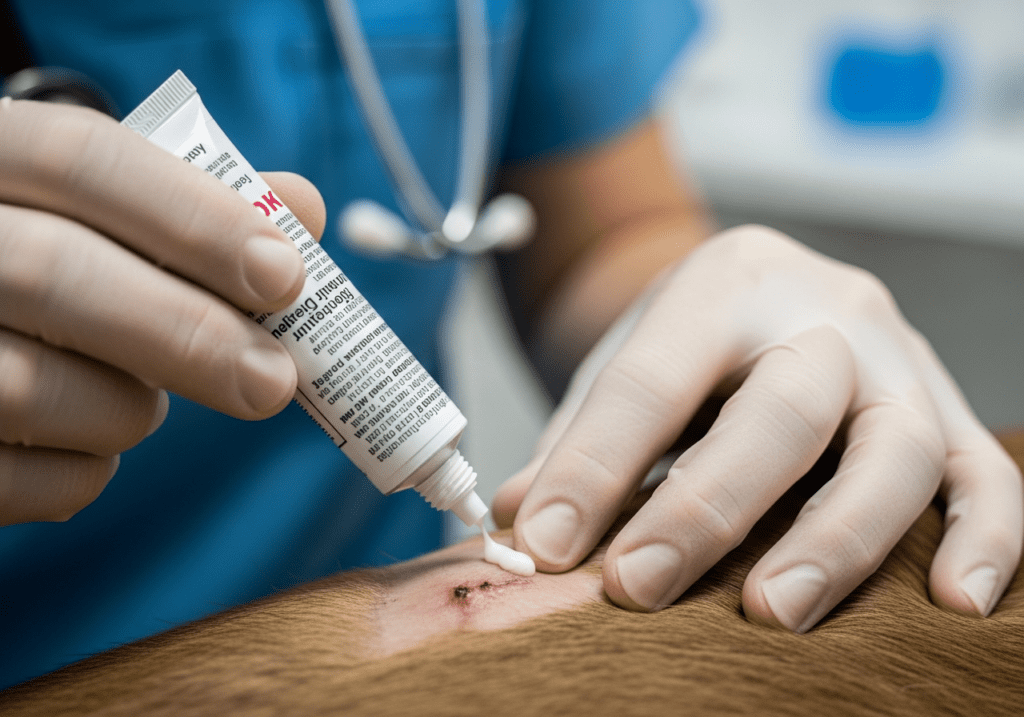
The recent FDA announcement that grants emergency use authorization (EUA) for an antiseptic ointment specifically designed to treat New World screwworm infestations marks a significant development in veterinary medicine. This urgent need for an effective screwworm treatment affects a wide range of animal species and poses pressing challenges for veterinarians across the United States.
Understanding New World Screwworm Infestations
New World screwworms are parasitic larvae that infest warm-blooded animals, including livestock and companion animals. These infestations can lead to severe tissue damage, pain, and, in many cases, death if left untreated. Veterinary clinics, therefore, find themselves on the front lines of managing these infestations, making it essential for them to understand the implications of this new screwworm treatment.
The Role of the Antiseptic Ointment
The newly authorized ointment, designed to act as an antiseptic barrier, provides a crucial line of defense against these infestations. By using this ointment, veterinary clinics can improve their patient care protocols significantly. For general practice veterinarians and urgent care veterinarians, having access to this treatment allows them to better address acute cases of screwworm infestations as they arise.
Clinical Implications for Veterinary Practices
Implementing this new screwworm treatment can enhance operational efficiency in veterinary clinics. Clinic owners and practice managers should consider training their staff on the proper application and medical management of animals affected by screwworms. Additionally, this training can serve as a valuable opportunity to educate the community about prevention and control measures.
Addressing Key Challenges
Veterinarians face the challenge of rapid identification and treatment of screwworm infestations, especially in emergency and critical care settings. Those working with food animals and mixed species face unique hurdles, given the scale of potential infections in livestock populations. Therefore, staying informed about the latest treatments, like the FDA’s new screwworm ointment, is vital for any veterinary professional tackling this urgent issue.
Anticipating Future Trends
As we look to the future, it’s important to consider the broader implications of regulatory updates like the recent FDA EUA. These interactions between veterinary medicine and regulatory bodies will likely shape treatment modalities moving forward. Understanding these trends will enable veterinary internal medicine specialists and shelter veterinarians to adapt their practices accordingly.
Conclusion
In conclusion, the FDA’s emergency use authorization for the new screwworm ointment equips veterinarians with a crucial tool in the fight against these devastating infestations. By integrating this treatment, veterinary clinics can enhance their patient care and operational efficiency. As the industry continues to evolve, having access to top talent becomes increasingly important. Pulivarthi Group stands ready to assist veterinary clinics in securing hard-to-find experts across various roles, including associate veterinarians, urgent care veterinarians, and board-certified specialists. Together, we can improve animal health outcomes and navigate the challenges ahead.